|
11/15/2023 0 Comments Co element formula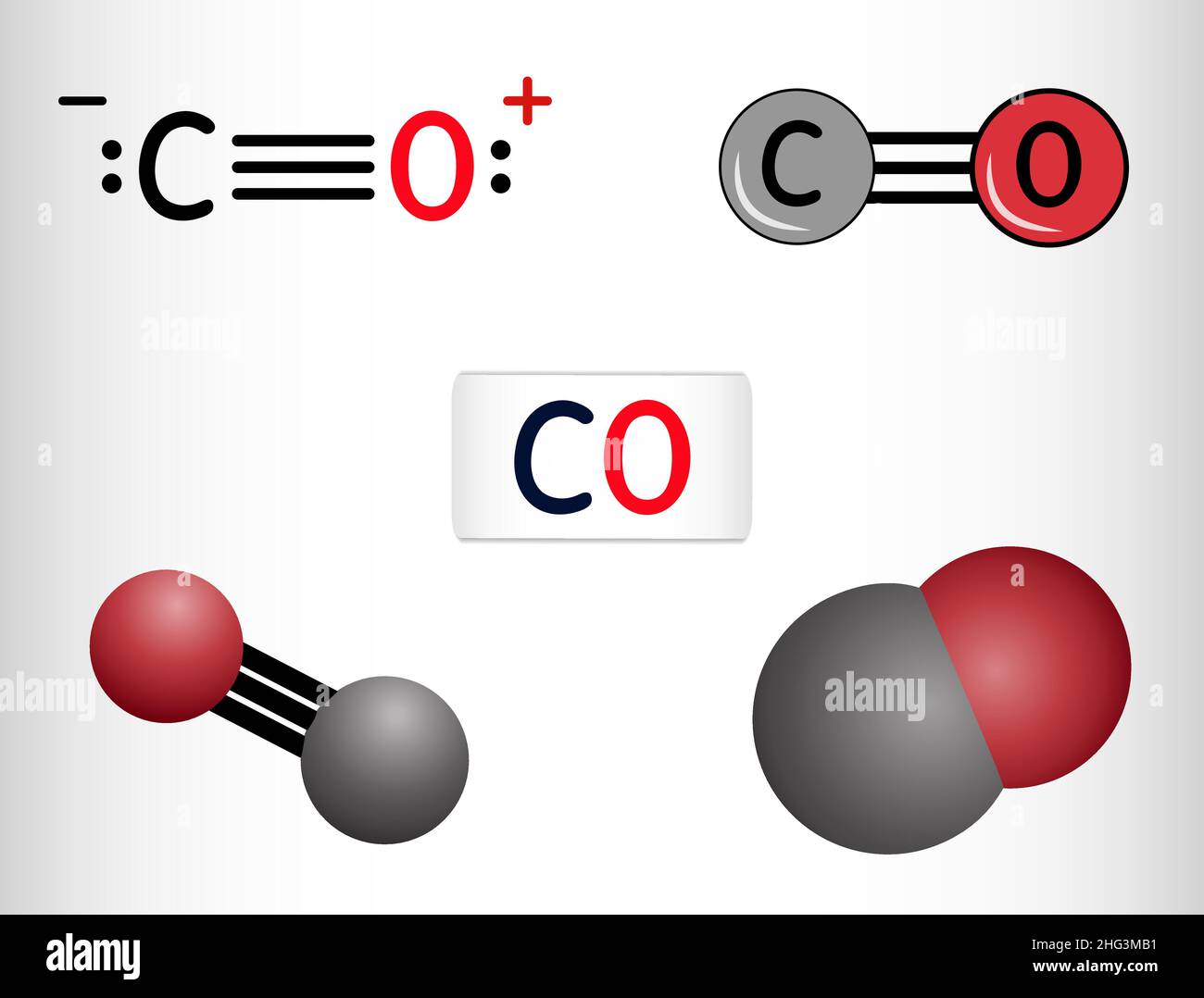
So the molecular weight of Carbon monoxide is 28.0101. Step 4: Calculate the molecular weight of Carbon monoxide by adding up the total weight of all atoms. Total weight of Oxygen atoms in Carbon monoxide: 15.9994 x 1 = 15.9994 Number of Oxygen atoms in Carbon monoxide: 1 Total weight of Carbon atoms in Carbon monoxide: 12.0107 x 1 = 12.0107 Number of Carbon atoms in Carbon monoxide: 1 Step 3: Calculate the total weight of each atom present in a Carbon monoxide molecule by multiplying its atomic weight by its number. Step 2: Find out atomic weights of each atom (from periodic table).Ītomic weight of Carbon (C): 12.0107 (Ref: Jlab-ele006)Ītomic weight of Oxygen (O) : 15.9994 (Ref: Jlab-ele008) From the chemical formula of Carbon monoxide, you can find that one molecule of Carbon monoxide has one Carbon (C) atom, and one Oxygen (O) atom. Chemical formula of Carbon monoxide is CO. Ca 10 (PO 4) 6 (OH) 2 Hydroxyapatite, that is present in the enamel of the tooth. C 18 H 21 NO 3 codeine, a painkilling drug. Step 1: Find out the chemical formula and determine constituent atoms and their number in a Carbon monoxide molecule.įrom the chemical formula, you will know different atoms and their number in a Carbon monoxide molecule. Problem 1: In one molecule of the compound, determine how many atoms of every element are present for each one of these chemical formulas. Note that the value of atomic weight may differ slightly from different sources.ĬALCULATION PROCEDURE: Carbon monoxide Molecular Weight Calculation The sum of total weight of all constituent atoms will be the molecular weight of the compound. 
Then calculate the total weight of each atom by multiplying its atomic weight by its number. To calculate molecular weight of any compound, the first step is to know the constituent atoms and their number in that particular compound. I'm a little confused because there are times where we do use this, but I am not sure how to identify when to use the total atomic masses combined.The molecular weight of Carbon monoxide is 28.0101. I want to understand why we don't use the atomic masses added together to divide and instead use the mass that is given. Classified as a transition metal, Cobalt is a solid at room temperature. I divided 65.98 g F/ 77.93 g/ mol = 0.85 x 100% = 85% and Co = 24.4 %. Cobalt is a chemical element with symbol Co and atomic number 27. When solving this problem, I added both 58.93 g/mol and 19.000 g/mol to get 77.93 g/mol and I used this number to find the mass % compositions. We are constantly adding new data and working. Find chemical and physical properties, biological activities, safety and toxicity information, patents, literature citations and more. Search chemicals by name, molecular formula, structure, and other identifiers. In this case you get 1 mole of cobalt to 6 moles of fluorine, and therefore the empirical formula is CoF6. PubChem is the world's largest collection of freely accessible chemical information. Next, divide all elements by the smallest amount of moles, in this case, divide both elements by the moles of cobalt. Now assuming 100 g sample, convert the elements to moles. Next you need to find the mass % of each element in the compound. And 339.20 grams of the compound is Cobalt, meaning the remaining 656.88 grams is Fluorine. Chem_Mod wrote:The total mass of the compound is 996.08 grams.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
